
School science visits since 2004!
– Curriculum-linked & award-winning incursions.
– Over 40 primary & high school programs to choose from.
– Designed by experienced educators.
– Over 2 million students reached.
– Face to face incursions & online programs available.
– Early learning centre visits too!

Get the Unit of Work on Light & Colour here!
- What is colour addition vs subtraction?
- What is opacity, translucence & transparency?
- What is the difference between refraction & reflection?
- Explore UV light, lenses, thermochromism and more!
Includes cross-curricular teaching ideas, student quizzes, a sample marking rubric, scope & sequences & more
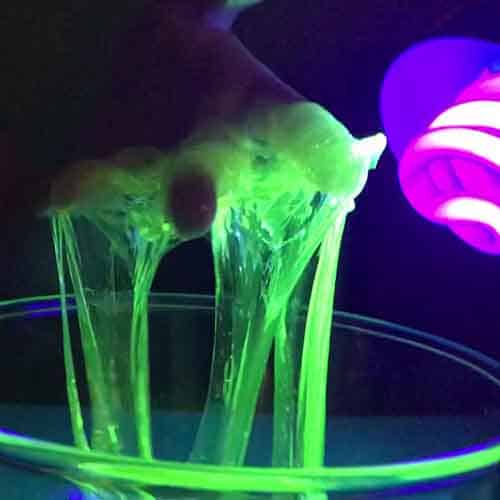
Why Does This Happen?
Visible light ranges in frequency from VIOLET (400nm) to RED (700nm).
Black lights emit the invisible ultraviolet light called UV-A, which has less energy than the sunburn causing UV-B light.
Some chemicals can absorb energy from invisible UV-A light and emit it as visible light.
These chemicals are said to be fluorescent and can occur in everyday items such as
laundry detergents, driver’s licences, air mail, nail polish, oils and even money!
All materials are made up of atoms, whereby electrons move in layered orbits or ‘shells’ around a central nucleus of protons and neutrons (except the nucleus of Hydrogen which contains only a proton). Fluorescence occurs when the UV-A light energizes the electrons surrounding the atoms that make up the paper. As the electrons get energized, they move to the outer orbit (or electron shell). Once the electrons lose their additional energy they fall back to their original orbit, releasing any remaining extra energy as visible light.
Forensic scientists can use fluorescence of chemicals to link a crime scene to the guilty person!
Variables to test
- Try different brands of petroleum jelly.
- What happens if you use different colours of paper?
From image formation to rainbow science, we’ve got your unit on light covered!
Get in touch with FizzicsEd to find out how we can work with your class.
Light & Sound
Years 1 to 6
Maximum 30 students
Workshop or Show (NSW & VIC)
60 minute show or 90 minute workshop
Online Class Available
Light & Sound
Years 1 to 6
Maximum 30 students
Workshop or Show (NSW & VIC)
60 minute show or 90 minute workshop
Online Class Available
STEM Full Day Accelerator - Primary
Designed from real classroom experiences, this modular day helps you create consistently effective science learning that directly address the new curriculum with easily accessible and cost-effective materials.




























Comments